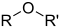
Funktionel gruppe
From Wikipedia, the free encyclopedia
I kemi er en funktionel gruppe den del af et molekyle som bestemmer molekylets kemiske egenskaber og dermed hvilke reaktioner molekylet kan deltage i. Et molekyle kan indeholde mere end en funktionel gruppe, eller slet ingen.
Flere oplysninger Klasse, Gruppe ...
| Klasse | Gruppe | Formel | Strukturformel | Præfiks | Suffiks | Eksempel |
|---|---|---|---|---|---|---|
| Acylhalid (syreklorid) | Haloformyl | RCOX |  |
haloformyl- | -oylhalid | Acetylklorid (Ethanoylklorid) |
| Alkohol | Hydroxyl | ROH | hydroxy- | -ol | 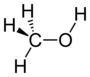 Methanol | |
| Aldehyd | Aldehyd | RCHO |  |
oxo- | -al | 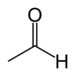 Acetaldehyd (Ethanal) |
| Alkan | Alkyl | RH | alkyl- | -an | 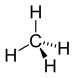 Methan | |
| Alken | Alkenyl | R2C=CR2 | 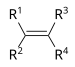 |
alkenyl- | -en |  Ethylen (Ethen) |
| Alkyn | Alkynyl | RC≡CR' | alkynyl- | -yn | Acetylen (Ethyn) | |
| Amid | Carboxamid | RCONR2 |  |
carboxamido- | -amid |  Acetamid (Ethanamid) |
| Aminer | Primær amin | RNH2 | amino- | -amin | 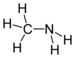 Metylamin (Methanamin) | |
| Sekundær amin | R2NH |  |
amino- | -amin | Dimethylamin | |
| Tertiær amin | R3N | 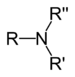 |
amino- | -amin | 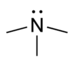 Trimethylamin | |
| Kvarternær ammoniumkation | R4N+ | 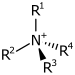 |
ammonio- | -ammonium |  Kolin | |
| Azoforbindelse | Azo (Diimid) |
RN2R' | azo- | -diazen | 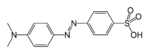 Methylorange (p-dimethylamino- azobenzensulfonsyre) | |
| Toluenderivater | Benzyl | RCH2C6H5 RBn |
benzyl- | 1-(substituent)toluen | Benzylbromid (1-Bromtoluen) | |
| Carbonat | Carbonatester | ROCOOR | alkyl carbonat | |||
| Carboxylat | Carboxylat | RCOO− | 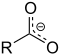 |
carboxy- | -oat | Natriumacetat (Natriumethanoat) |
| Carboxylsyre | Carboxyl | RCOOH | 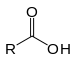 |
carboxy- | -(carboxyl)syre | 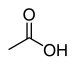 Eddikesyre (Ethansyre) |
| Cyanater | Cyanat | ROCN | cyanato- | alkyl cyanat | ||
| Thiocyanat | RSCN | thiocyanato- | alkyl thiocyanat | |||
| Æter | Æter | ROR' | alkoxy- | alkyl-alkyl-æter | Diætylæter (Ethoxyethan) | |
| Ester | Ester | RCOOR' | alkyl-alkanoate | Ætylbutyrat (Ætylbutanoat) | ||
| Haloalkan | Halo | RX |  |
halo- | alkylhalid | Chlorethan (Ætylklorid) |
| Hydroperoxid | Hydroperoxy | ROOH | hydroperoxy- | alkylhydroperoxid | Methylethylketonperoxid | |
| Imin | Primær ketimin | RC(=NH)R' |  |
imino- | -imin | |
| Sekundær ketimin | RC(=NR)R' |  |
imino- | -imin | ||
| Primær aldimin | RC(=NH)H |  |
imino- | -imin | ||
| Sekundær aldimin | RC(=NR')H | 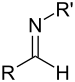 |
imino- | -imin | ||
| Isocyanid | Isocyanid | RNC | isocyano- | alkylisocyanid | ||
| Isocyanater | Isocyanat | RNCO | isocyanato- | alkylisocyanat | Methylisocyanat | |
| Isothiocyanat | RNCS | isothiocyanato- | alkylisothiocyanat | Allylisothiocyanat | ||
| Keton | Carbonyl | RCOR' | 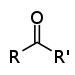 |
keto-, oxo- | -on | 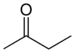 Methylethylketon (Butanon) |
| Nitrat | Nitrat | RONO2 | nitrooxy-, nitroxy- |
alkylnitrat |
 Amylnitrat (1-nitrooxypentan) | |
| Nitril | Nitril | RCN | cyano- |
alkannitril |
 Benzonitril (Phenylcyanid) | |
| Nitrit | Nitrosooxy | RONO | nitrosooxy- |
alkylnitrit |
 Amylnitrit (3-methyl-1-nitrosooxybutan) | |
| Nitroforbindelse | Nitro | RNO2 | 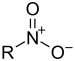 |
nitro- | 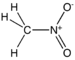 Nitromethan | |
| Nitrosoforbindelse | Nitroso | RNO | nitroso- | Nitrosobenzen | ||
| Peroxid | Peroxy | ROOR | peroxy- | alkylperoxid | Di-tert-butylperoxid | |
| Benzenderivater | Phenyl | RC6H5 | phenyl- | -benzen | 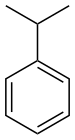 Cumene (2-phenylpropan) | |
| Fosfin | Fosfino | R3P | phosphino- | -phosphan | Methylpropylphosphan | |
| Fosfodiester | Fosfat | HOPO(OR)2 | fosforsyre- di(substituent)ester |
di(substituent)- hydrogenfosfat |
DNA | |
| Phosphosyre | Phosphono | RP(=O)(OH)2 | 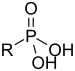 |
phosphono- | substituent phosphonsyre | Benzylphosphonic acid |
| Fosfat | Fosfat | ROP(=O)(OH)2 |  |
phospho- | Glyceraldehyd-3-fosfat | |
| Pyridinderivater | Pyridyl | RC5H4N |
4-pyridyl |
-pyridin |  Nicotin | |
| Sulfid | RSR' | di(substituent)sulfid | Dimethylsulfid | |||
| Sulfon | Sulfonyl | RSO2R' |  |
sulfonyl- | di(substituent)- sulfon |
Dimethylsulfon (Metylsulfonylmetan) |
| Sulfonsyre | Sulfo | RSO3H |  |
sulfo- | substituent- sulfonsyre |
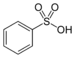 Benzensulfonsyre |
| Sulfoxid | Sulfinyl | RSOR' | 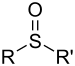 |
sulfinyl- | di(substituent)- sulfoxid |
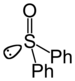 Difenylsulfoxid |
| Thiol | Sulfhydryl | RSH | mercapto-, sulfanyl- | -thiol | Ethanthiol (Ethylmercaptan) |
Luk
| | Spire Denne artikel om kemi er en spire som bør udbygges. Du er velkommen til at hjælpe Wikipedia ved at udvide den. |