Chromate and dichromate
Chromium(VI) anions / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Dichromate?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
This article is about the salts of the chromium(VI) anion. For other uses, see Chromate. See also for disambiguation of derived terms.
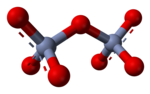
Chromate salts contain the chromate anion, CrO2−
4. Dichromate salts contain the dichromate anion, Cr
2O2−
7. They are oxyanions of chromium in the +6 oxidation state and are moderately strong oxidizing agents. In an aqueous solution, chromate and dichromate ions can be interconvertible.
Quick Facts Names, Identifiers ...
| |||
| |||
| Names | |||
|---|---|---|---|
| Systematic IUPAC name
Chromate and dichromate | |||
| Identifiers | |||
| |||
3D model (JSmol) |
| ||
| ChEBI |
| ||
| DrugBank |
| ||
PubChem CID |
|||
| UNII |
| ||
CompTox Dashboard (EPA) |
| ||
| |||
| |||
| Properties | |||
| CrO2− 4 and Cr 2O2− 7 | |||
| Molar mass | 115.994 g mol−1 and 215.988 g mol−1 | ||
| Conjugate acid | Chromic acid | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Close