
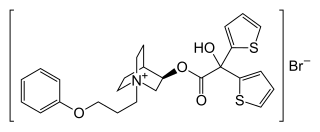
Aclidinium bromide
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Aclidinium bromide?
Summarize this article for a 10 year old
Aclidinium bromide (INN) is a long-acting, inhaled muscarinic antagonist (LAMA) approved in the United States on July 24, 2012[2] as a maintenance treatment for chronic obstructive pulmonary disease (COPD).[3]
 | |
| Clinical data | |
|---|---|
| Trade names | Bretaris Genuair, Eklira Genuair, Tudorza Pressair |
| AHFS/Drugs.com | Monograph |
| License data |
|
| Routes of administration | Inhalation |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | <5% (in system) 30% (in lung) |
| Metabolism | Ester hydrolysis |
| Elimination half-life | 2–3 hrs |
| Duration of action | >24 hrs |
| Excretion | 65% urine, 33% feces |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.260.213 |
| Chemical and physical data | |
| Formula | C26H30BrNO4S2 |
| Molar mass | 564.55 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Evidence shows that it can improve quality of life and prevent hospitalization in those with COPD.[4] However, it does not appear to affect the risk of death or the frequency steroids are needed.[4] It is unclear if it differs from the similar medication tiotropium or other commonly used medications from the class of LAMAs.[4]
Aclidinium is delivered via a multidose dry powder inhaler, the Genuair inhaler. It is on the World Health Organization's List of Essential Medicines.[5]