BSTFA
Chemical compound / From Wikipedia, the free encyclopedia
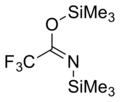
N,O-Bis(trimethylsilyl)trifluoroacetamide (BSTFA) is an organosilicon compound. It is a colorless liquid that is very sensitive to traces of water or alcohols.
Quick Facts Names, Identifiers ...
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
trimethylsilyl 2,2,2-trifluoro-N-trimethylsilylethanimidate | |||
| Other names
BSTFA, N,O-Bis(trimethylsilyl)trifluoroacetamide | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChEBI | |||
| ChemSpider |
| ||
| ECHA InfoCard | 100.042.807 | ||
PubChem CID |
|||
| |||
| |||
| Properties | |||
| C8H18F3NOSi2 | |||
| Molar mass | 257.403 g·mol−1 | ||
| Appearance | colourless liquid | ||
| Density | 0.96 | ||
| Melting point | −10 °C (14 °F; 263 K) | ||
| Boiling point | 45–55 °C (113–131 °F; 318–328 K) 14 mm Hg | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Close
It is often used to convert hydroxyl groups to trimethylsilyl ether groups (Me = CH3):
- ROH + CF3C(OSiMe3)NSiMe3 → CF3C(O)NH(SiMe3) + ROSiMe3
These silylated derivatives are amenable to analysis or further manipulation. Siloxanes are invariably more volatile than their hydroxyl precursors, and thus they can be more easily analyzed with gas chromatography.[1]
This reagent was first reported in 1968.[2]