
Chromium(II) acetate
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Chromium(II) acetate?
Summarize this article for a 10 year old
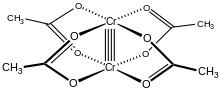
Chromium(II) acetate hydrate, also known as chromous acetate, is the coordination compound with the formula Cr2(CH3CO2)4(H2O)2. This formula is commonly abbreviated Cr2(OAc)4(H2O)2. This red-coloured compound features a quadruple bond. The preparation of chromous acetate once was a standard test of the synthetic skills of students due to its sensitivity to air and the dramatic colour changes that accompany its oxidation. It exists as the dihydrate and the anhydrous forms.
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Chromium(II) acetate hydrate | |
| Other names
chromous acetate, chromium diacetate, chromium(II) ethanoate | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.224.848 |
PubChem CID |
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C8H16Cr2O10 | |
| Molar mass | 376.198 g·mol−1 |
| Appearance | brick-red solid |
| Density | 1.79 g/cm3 |
| Melting point | dehydrates |
| soluble in hot water, MeOH | |
| -5104.0·10−6 cm3/mol | |
| Structure | |
| monoclinic | |
| octahedral counting the Cr–Cr bond | |
| 0 D | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
could react exothermically in air |
| Related compounds | |
Related compounds |
Rh2(OAc)4(H2O)2 Cu2(OAc)4(H2O)2, molybdenum(II) acetate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Cr2(OAc)4(H2O)2 is a reddish diamagnetic powder, although diamond-shaped tabular crystals can be grown. Consistent with the fact that it is nonionic, Cr2(OAc)4(H2O)2 exhibits poor solubility in water and methanol.
