PFP (enzyme)
From Wikipedia, the free encyclopedia
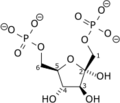
Diphosphate—fructose-6-phosphate 1-phosphotransferase also known as PFP is an enzyme of carbohydrate metabolism in plants and some bacteria. The enzyme (EC 2.7.1.90) catalyses the reversible interconversion of fructose 6-phosphate and fructose 1,6-bisphosphate using inorganic pyrophosphate as the phosphoryl donor:
- diphosphate + D-fructose 6-phosphate
phosphate + D-fructose 1,6-bisphosphate
| Diphosphate—fructose-6-phosphate 1-phosphotransferase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 Bacillus stearothermophilus phosphofructokinase.[1] | |||||||||
| Identifiers | |||||||||
| EC no. | 2.7.1.90 | ||||||||
| CAS no. | 55326-40-4 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
In plants, the PFP is located in the cytosol of the cell and is strongly activated by the signal molecule fructose 2,6-bisphosphate.
PFP is an exclusively cytosolic enzyme that catalyses the phosphorylation of fructose-6-phosphate to fructose-1,6-bisphosphate in the glycolytic direction, and the de-phosphorylation of fructose-1,6-bisphosphate to fructose-6-phosphate in the gluconeogenic reaction. Reeves[2] first isolated PFP from Entamoeba histolytica, a lower eukaryote. The first plant PFP isolated was from the leaves of pineapples by Carnal and Black[3] and it has since been isolated from a variety of plant species and tissues.[4]