
Ebola
Viral hemorrhagic fever of humans and other primates caused by ebolaviruses / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Ebola virus disease?
Summarize this article for a 10 year old
Ebola, also known as Ebola virus disease (EVD) and Ebola hemorrhagic fever (EHF), is a viral hemorrhagic fever in humans and other primates, caused by ebolaviruses.[1] Symptoms typically start anywhere between two days and three weeks after infection.[3] The first symptoms are usually fever, sore throat, muscle pain, and headaches.[1] These are usually followed by vomiting, diarrhoea, rash and decreased liver and kidney function,[1] at which point some people begin to bleed both internally and externally.[1] It kills between 25% and 90% of those infected – about 50% on average.[1] Death is often due to shock from fluid loss, and typically occurs between six and 16 days after the first symptoms appear.[2] Early treatment of symptoms increases the survival rate considerably compared to late start.[4] An Ebola vaccine was approved by the US FDA in December 2019.
| Ebola | |
|---|---|
| Other names | Ebola haemorrhagic fever (EHF), Ebola virus disease |
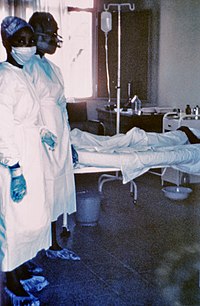 | |
| Two nurses standing near Mayinga N'Seka, a nurse with Ebola virus disease in the 1976 outbreak in Zaire. N'Seka died a few days later. | |
| Specialty | Infectious disease |
| Symptoms | Fever, sore throat, muscular pain, headaches, diarrhoea, bleeding[1] |
| Complications | shock from fluid loss[2] |
| Usual onset | Two days to three weeks post exposure[1] |
| Causes | Ebolaviruses spread by direct contact[1] |
| Diagnostic method | Finding the virus, viral RNA, or antibodies in blood[1] |
| Differential diagnosis | Malaria, cholera, typhoid fever, meningitis, other viral haemorrhagic fevers[1] |
| Prevention | Coordinated medical services, careful handling of bushmeat[1] |
| Treatment | Supportive care[1] |
| Medication | Atoltivimab/maftivimab/odesivimab (Inmazeb) |
| Prognosis | 25–90% mortality[1] |
The virus spreads through direct contact with body fluids, such as blood from infected humans or other animals,[1] or from contact with items that have recently been contaminated with infected body fluids.[1] There have been no documented cases, either in nature or under laboratory conditions, of spread through the air between humans or other primates.[5] After recovering from Ebola, semen or breast milk may continue to carry the virus for anywhere between several weeks to several months.[1][6][7] Fruit bats are believed to be the normal carrier in nature; they are able to spread the virus without being affected by it.[1] The symptoms of Ebola may resemble those of several other diseases, including malaria, cholera, typhoid fever, meningitis and other viral hemorrhagic fevers.[1] Diagnosis is confirmed by testing blood samples for the presence of viral RNA, viral antibodies or the virus itself.[1][8]
Control of outbreaks requires coordinated medical services and community engagement,[1] including rapid detection, contact tracing of those exposed, quick access to laboratory services, care for those infected, and proper disposal of the dead through cremation or burial.[1][9] Prevention measures involve wearing proper protective clothing and washing hands when in close proximity to patients and while handling potentially infected bushmeat, as well as thoroughly cooking bushmeat.[1] An Ebola vaccine was approved by the US FDA in December 2019.[10] While there is no approved treatment for Ebola as of 2019[update],[11] two treatments (atoltivimab/maftivimab/odesivimab and ansuvimab) are associated with improved outcomes.[12] Supportive efforts also improve outcomes.[1] These include oral rehydration therapy (drinking slightly sweetened and salty water) or giving intravenous fluids, and treating symptoms.[1] In October 2020, atoltivimab/maftivimab/odesivimab (Inmazeb) was approved for medical use in the United States to treat the disease caused by Zaire ebolavirus.[13]