
Enantiomer
Stereoisomers that are nonsuperposable mirror images of each other / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Enantiomers?
Summarize this article for a 10 year old
In chemistry, an enantiomer (/ɪˈnænti.əmər, ɛ-, -oʊ-/[1] ih-NAN-tee-ə-mər; from Ancient Greek ἐναντίος (enantíos) 'opposite', and μέρος (méros) 'part') – also called optical isomer,[2] antipode,[3] or optical antipode[4] – is one of two stereoisomers that are nonsuperposable onto their own mirror image. Enantiomers are much like one's right and left hands; without mirroring one of them, hands cannot be superposed onto each other.[5] No amount of reorientation in three spatial dimensions will allow the four unique groups on the chiral carbon (see chirality) to line up exactly. The number of stereoisomers a molecule has can be determined by the number of chiral carbons it has.
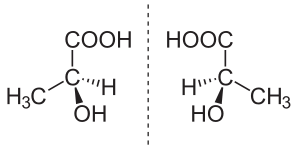
A molecule with chirality rotates plane-polarized light.[6] A mixture of equal amounts of each enantiomer, a racemic mixture or a racemate, does not rotate light.[7][8] [9]
Stereoisomers include both enantiomers and diastereomers. Diastereomers, like enantiomers, share the same molecular formula and are also nonsuperposable onto each other; however, they are not mirror images of each other.[10]