
Estradiol/megestrol acetate
Pharmaceutical combination / From Wikipedia, the free encyclopedia
Estradiol/megestrol acetate (E2/MGA), sold under the brand names Mego-E and Chinese injectable No. 2, is a form of combined injectable birth control which is used in the People's Republic of China.[1][2][3][4][5][6][7] It contains 3.5 mg estradiol (E2), an estrogen, and 25 mg megestrol acetate (MGA), a progestin.[3][4][5] It is a microcrystalline aqueous suspension with a defined particle size range.[3][4][5] The medication is given once per month by injection into muscle.[3][4][5]
 | |
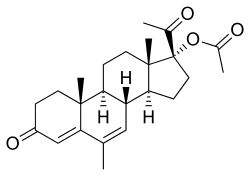 | |
| Combination of | |
|---|---|
| Estradiol | Estrogen |
| Megestrol acetate | Progestogen |
| Clinical data | |
| Trade names | Mego-E, Chinese injectable No. 2 |
| Other names | E2/MGA |
| Routes of administration | Intramuscular injection |
| Identifiers | |
| CAS Number | |
Studies of E2/MGA have been published.[8][9][10][11][12][13][14][15][16][17] The elimination half-life of MGA in this formulation is 14.35 ± 9.1 days.[10] The plasma protein binding of MGA to albumin is 82.4%, while none is bound to sex hormone-binding globulin.[10] Following an injection of E2/MGA, estradiol increased after 24 hours and persisted at high levels for 5 days, thereafter decreasing to low levels.[13]
In 1969, MGA was studied in China as an aqueous suspension for parenteral administration in animals.[18] The next year, it was studied in women as a progestogen-only injectable contraceptive, with a dosing interval of once every 3 months by intramuscular injection.[18] It was effective as a contraceptive but was associated with menstrual irregularities.[18] Starting in 1973, a combination of estradiol cypionate (EC) and MGA was studied in women as a combined injectable contraceptive over a period of 3 years.[18][1][6][2][19] E2/MGA, an "improvement" of EC/MGA, was studied in China in large clinical trials from 1977 to 1979 and was approved for use in this country in 1980.[1][6][12] By 1987, production of E2/MGA had reached 9 million units per year and had spread to over 22 Chinese provinces and cities.[12] E2/MGA appears to have been discontinued sometime between 2005 and 2008.[20]
A follow-up product consisting of 5 mg estradiol valerate (EV) and 15 mg MGA encapsulated in 50 to 80 μm-diameter microspheres as an aqueous suspension for use by intramuscular injection was developed and studied in China as well but was never marketed.[4][21][22][23][24][25][26] Following an injection, levels of MGA were higher than 2 ng/mL after a day, reached a peak of 3.2 ng/mL after 8 days, remained at levels of 2 ng/mL after 27 days, remained at 1 to 2 ng/mL after 27 to 45 days, and were below 1 ng/mL after 45 to 51 days (0.71 ng/mL on the 51st day).[26]