
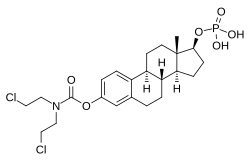
Estramustine phosphate
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Estramustine phosphate?
Summarize this article for a 10 year old
Estramustine phosphate (EMP), also known as estradiol normustine phosphate and sold under the brand names Emcyt and Estracyt, is a dual estrogen and chemotherapy medication which is used in the treatment of prostate cancer in men.[7][5][8][9][10][3][1][11][6][12] It is taken multiple times a day by mouth or by injection into a vein.[7][8][3][1][6][12]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Emcyt, Estracyt |
| Other names | EMP; Leo 299; NSC-89199; Ro 21-8837/001; Estradiol normustine phosphate; Estradiol 3-normustine 17β-phosphate; Estradiol 3-(bis(2-chloroethyl)carbamate) 17β-(dihydrogen phosphate) |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a608046 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth, IV |
| Drug class | Chemotherapeutic agent; Estrogen; Estrogen ester |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 44–75% (as estramustine and estromustine)[1] |
| Protein binding | • Estradiol: 98%[2] • Estrone: 96%[2] |
| Metabolism | Liver, intestines[3][1][4] |
| Metabolites | • Estramustine[3][1] • Estromustine[3][1] • Estradiol[3][1] • Estrone[3][1] • Phosphoric acid[3][1] • Normustine[5] |
| Elimination half-life | • EMP: 1.27 hours[6] • Estromustine: 10–14 hrs[1] • Estrone: 15–17 hours[1] |
| Excretion | Bile, feces (2.9–4.8%)[1][4] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.023.193 |
| Chemical and physical data | |
| Formula | C23H32Cl2NO6P |
| Molar mass | 520.38 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Side effects of EMP include nausea, vomiting, gynecomastia, feminization, demasculinization, sexual dysfunction, blood clots, and cardiovascular complications.[3][9][13] EMP is a dual cytostatic and hence chemotherapeutic agent and a hormonal anticancer agent of the estrogen type.[1][3][14][6] It is a prodrug of estramustine and estromustine in terms of its cytostatic effects and a prodrug of estradiol in relation to its estrogenic effects.[1][3] EMP has strong estrogenic effects at typical clinical dosages, and consequently has marked antigonadotropic and functional antiandrogenic effects.[5][1][3][14]
EMP was introduced for medical use in the early 1970s.[3] It is available in the United States, Canada, the United Kingdom, other European countries, and elsewhere in the world.[15][16]