
Gallium(III) oxide
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Gallium(III) oxide?
Summarize this article for a 10 year old
Gallium(III) oxide is an inorganic compound and ultra-wide-bandgap semiconductor with the formula Ga2O3. It is actively studied for applications in power electronics, phosphors, and gas sensing.[5][6][7] The compound has several polymorphs, of which the monoclinic β-phase is the most stable. The β-phase’s bandgap of 4.7–4.9 eV and large-area, native substrates make it a promising competitor to GaN and SiC-based power electronics applications and solar-blind UV photodetectors.[7][8] The orthorhombic ĸ-Ga2O3 is the second most stable polymorph. The ĸ-phase has shown instability of subsurface doping density under thermal exposure.[9] Ga2O3 exhibits reduced thermal conductivity and electron mobility by an order of magnitude compared to GaN and SiC, but is predicted to be significantly more cost-effective due to being the only wide-bandgap material capable of being grown from melt.[7][10][11] β-Ga2O3 is thought to be radiation-hard, which makes it promising for military and space applications.[12][13]
 β-Ga2O3 crystal | |
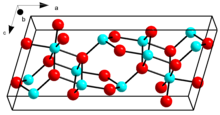 Crystal structure of β-Ga2O3 | |
| Names | |
|---|---|
| Other names
gallium trioxide, gallium sesquioxide | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.031.525 |
| EC Number |
|
PubChem CID |
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| Ga2O3 | |
| Molar mass | 187.444 g/mol |
| Appearance | white crystalline powder |
| Melting point | 1,725 °C (3,137 °F; 1,998 K)[1] |
| insoluble | |
| Solubility | soluble in most acids |
| Structure[2][3] | |
| Monoclinic, mS20, space group = C2/m, No. 12 | |
a = 1.2232 nm, b = 0.3041 nm, c = 0.5801 nm α = 90°, β = 103.73°, γ = 90° β-phase | |
Formula units (Z) |
4 |
| Thermochemistry[4] | |
Heat capacity (C) |
92.1 J/(mol·K) |
Std molar entropy (S⦵298) |
85.0 J/(mol·K) |
Std enthalpy of formation (ΔfH⦵298) |
−1089.1 kJ/mol |
Gibbs free energy (ΔfG⦵) |
−998.3 kJ/mol |
Enthalpy of fusion (ΔfH⦵fus) |
100 kJ/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |