γ-Carotene
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Gamma-Carotene?
Summarize this article for a 10 year old
γ-Carotene (gamma-carotene) is a carotenoid, and is a biosynthetic intermediate for cyclized carotenoid synthesis in plants.[2] It is formed from cyclization of lycopene by lycopene cyclase epsilon.[3] Along with several other carotenoids, γ-carotene is a vitamer of vitamin A in herbivores and omnivores. Carotenoids with a cyclized, beta-ionone ring can be converted to vitamin A, also known as retinol, by the enzyme beta-carotene 15,15'-dioxygenase; however, the bioconversion of γ-carotene to retinol has not been well-characterized. γ-Carotene has tentatively been identified as a biomarker for green and purple sulfur bacteria in a sample from the 1.640 ± 0.003-Gyr-old Barney Creek Formation in Northern Australia which comprises marine sediments.[2] Tentative discovery of γ-carotene in marine sediments implies a past euxinic environment, where water columns were anoxic and sulfidic.[2] This is significant for reconstructing past oceanic conditions, but so far γ-carotene has only been potentially identified in the one measured sample.
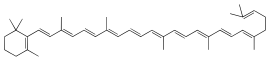 | |
 | |
| Names | |
|---|---|
| IUPAC name
β,ψ-Carotene | |
| Systematic IUPAC name
2-[(1E,3E,5E,7E,9E,11E,13E,15E,17E,19E)-3,7,12,16,20,24-Hexamethylpentacosa-1,3,5,7,9,11,13,15,17,19,23-undecaen-1-yl]-1,3,3-trimethylcyclohex-1-ene | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C40H56 | |
| Molar mass | 536.888 g·mol−1 |
| Melting point | 160 to 162 °C (320 to 324 °F; 433 to 435 K)[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |