
Goserelin
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Goserelin?
Summarize this article for a 10 year old
Goserelin, sold under the brand name Zoladex among others, is a medication which is used to suppress production of the sex hormones (testosterone and estrogen), particularly in the treatment of breast and prostate cancer.[1][2] It is an injectable gonadotropin releasing hormone agonist (GnRH agonist).
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Zoladex, others |
| Other names | D-Ser(But)6Azgly10-GnRH |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a601002 |
| Pregnancy category |
|
| Routes of administration | Implant |
| Drug class | GnRH analogue; GnRH agonist; Antigonadotropin |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | 27.3% |
| Elimination half-life | 4–5 hours |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.212.024 |
| Chemical and physical data | |
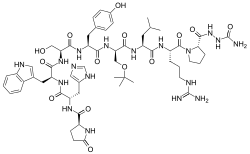
| Formula | C59H84N18O14 |
| Molar mass | 1269.433 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Structurally, it is a decapeptide. It is the natural GnRH decapeptide with two substitutions to inhibit rapid degradation.
Goserelin stimulates the production of the sex hormones testosterone and estrogen in a non-pulsatile (non-physiological) manner. This causes the disruption of the endogenous hormonal feedback systems, resulting in the down-regulation of testosterone and estrogen production.
It was patented in 1976 and approved for medical use in 1987.[3] It is on the World Health Organization's List of Essential Medicines.[4]