
Letrozole
Breast cancer drug / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Letrozole?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Letrozole, sold under the brand name Femara among others, is an aromatase inhibitor medication that is used in the treatment of breast cancer.[1]
Quick Facts Clinical data, Trade names ...
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Femara, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a698004 |
| License data | |
| Routes of administration | By mouth |
| Drug class | Aromatase inhibitor; Antiestrogen |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 99.9% |
| Protein binding | 60%, mainly to albumin |
| Metabolism | pharmacologically-inactive metabolites Bis(4-cyanophenyl)methanol and 4,4'-dicyanobenzophenone.[3] |
| Elimination half-life | 2 days[3] |
| Excretion | Kidney[3] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.200.357 |
| Chemical and physical data | |
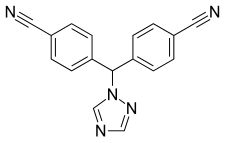
| Formula | C17H11N5 |
| Molar mass | 285.310 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Close
It was patented in 1986 and approved for medical use in 1996.[4] In 2021, it was the 222nd most commonly prescribed medication in the United States, with more than 1 million prescriptions.[5][6] It is on the World Health Organization's List of Essential Medicines.[7]