
Noretynodrel
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Noretynodrel?
Summarize this article for a 10 year old
Noretynodrel, or norethynodrel, sold under the brand name Enovid among others, is a progestin medication which was previously used in birth control pills and in the treatment of gynecological disorders but is now no longer marketed.[2][6][7][8] It was available both alone and in combination with an estrogen.[7][8][9] The medication is taken by mouth.[7]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Enovid (with mestranol), others |
| Other names | Norethynodrel; Noretinodrel Norethinodrel; NYD; SC-4642; NSC-15432; 5(10)-Norethisterone; 17α-Ethinyl-19-nor-5(10)-testosterone; 17α-Ethynyl-δ5(10)-19-nortestosterone; 17α-Ethynylestr-5(10)-en-17β-ol-3-one; 19-Nor-17α-pregn-5(10)-en-20-yn-17β-ol-3-one |
| Routes of administration | By mouth |
| Drug class | Progestogen; Progestin; Estrogen |
| ATC code | |
| Pharmacokinetic data | |
| Protein binding | Noretynodrel: to albumin and not to SHBGTooltip sex hormone-binding globulin or CBGTooltip corticosteroid-binding globulin[1] |
| Metabolism | Liver, intestines (hydroxylation, isomerization, conjugation)[1][2] |
| Metabolites | • 3α-Hydroxynoretynodrel[3] • 3β-Hydroxynoretynodrel[3] • Norethisterone[3][1][2] • Ethinylestradiol[2][4]• Conjugates[2] |
| Elimination half-life | Very short (< 30 minutes)[5] |
| Excretion | Breast milk: 1%[6] |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.000.620 |
| Chemical and physical data | |
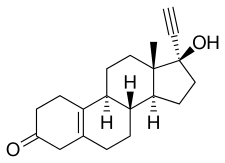
| Formula | C20H26O2 |
| Molar mass | 298.426 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Noretynodrel is a progestin, or a synthetic progestogen, and hence is an agonist of the progesterone receptor, the biological target of progestogens like progesterone.[2] It is a relatively weak progestogen.[10] The medication has weak estrogenic activity, no or only very weak androgenic activity, and no other important hormonal activity.[2][8][11][12] It is a prodrug of various active metabolites in the body, such as norethisterone among others.[2][13]
Noretynodrel was introduced for medical use in 1957.[8] It was specifically approved at this time in combination with mestranol for the treatment of gynecological and menstrual disorders.[8] Subsequently, in 1960, this formulation was approved for use as a birth control pill.[8][14] It was the first birth control pill to be introduced, and was followed by birth control pills containing norethisterone and other progestins shortly thereafter.[8][14][15] Due to its nature as a relatively weak progestogen, noretynodrel is no longer used in medicine.[10] As such, it is no longer marketed.[6][16]