
Octaoxygen
Allotrope of oxygen / From Wikipedia, the free encyclopedia
Octaoxygen, also known as ε-oxygen or red oxygen, is an allotrope of oxygen consisting of eight oxygen atoms. This allotrope forms above 600 K (327 °C; 620 °F) at pressures greater than 17 GPa.[1]
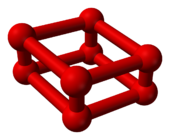 |  |
Ball-and-stick model of O8 | Part of the crystal structure of ε-oxygen |