
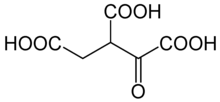
Oxalosuccinic acid
Chemical compound / From Wikipedia, the free encyclopedia
Oxalosuccinic acid is a substrate of the citric acid cycle. It is acted upon by isocitrate dehydrogenase. Salts and esters of oxalosuccinic acid are known as oxalosuccinates.
This article needs additional citations for verification. (May 2019) |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
1-Oxopropane-1,2,3-tricarboxylic acid | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider |
|
| ECHA InfoCard | 100.230.021 |
| KEGG |
|
PubChem CID |
|
| UNII | |
| |
| |
| Properties | |
| C6H6O7 | |
| Molar mass | 190.108 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Oxalosuccinic acid/oxalosuccinate is an unstable 6-carbon intermediate in the tricarboxylic acid cycle. It's a keto acid, formed during the oxidative decarboxylation of isocitrate to alpha-ketoglutarate, which is catalyzed by the enzyme isocitrate dehydrogenase. Isocitrate is first oxidized by coenzyme NAD+ to form oxalosuccinic acid/oxalosuccinate.[1] Oxalosuccinic acid is both an alpha-keto and a beta-keto acid (an unstable compound) and it is the beta-ketoic property that allows the loss of carbon dioxide in the enzymatic reaction in conversion to the five-carbon molecule 2-oxoglutarate.[2]