Loading AI tools
Chemical compound From Wikipedia, the free encyclopedia
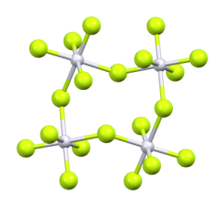
Ruthenium pentafluoride is the inorganic compound with the empirical formula RuF5. This green volatile solid has rarely been studied but is of interest as a binary fluoride of ruthenium, i.e. a compound containing only Ru and F. It is sensitive toward hydrolysis. Its structure consists of Ru4F20 tetramers, as seen in the isostructural platinum pentafluoride. Within the tetramers, each Ru adopts octahedral molecular geometry, with two bridging fluoride ligands.[1]
 | |
 | |
| Names | |
|---|---|
| IUPAC name
ruthenium(V) fluoride | |
| Other names
Ruthenium(V) fluoride, Ruthenium(5+) pentafluoride | |
| Identifiers | |
3D model (JSmol) |
|
| ECHA InfoCard | 100.035.015 |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| F5Ru | |
| Molar mass | 196.06 g/mol |
| Appearance | green solid |
| Density | 3.82 g/cm3 |
| Melting point | 86.5 °C (187.7 °F; 359.6 K) |
| Boiling point | 227 °C (441 °F; 500 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Ruthenium pentafluoride reacts with iodine to give ruthenium(III) fluoride.[2][3]
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.