
Silver(III) fluoride
Unstable silver compound in the unusual +3 oxidation state / From Wikipedia, the free encyclopedia
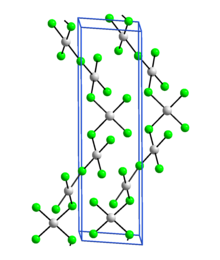
Silver(III) fluoride, AgF3, is an unstable, bright-red, diamagnetic compound containing silver in the unusual +3 oxidation state. Its crystal structure is very similar to that of gold(III) fluoride: it is a polymer consisting of rectangular AgF4 units linked into chains by fluoro bridges.[1]
Quick Facts Names, Identifiers ...
 | |
| Names | |
|---|---|
| IUPAC name
Silver(III) fluoride | |
| Other names
Silver trifluoride Argentic fluoride | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| 100808 | |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| AgF3 | |
| Molar mass | 164.86 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close