Loading AI tools
Class of chemical compounds containing an S=N bond From Wikipedia, the free encyclopedia
In chemistry, a sulfilimine (or sulfimide) is a type of chemical compound containing a sulfur-to-nitrogen bond which is often represented as a double bond (S=N). In fact, a double bond violates the octet rule, and the bond may be considered a single bond with a formal charge of +1 on the sulfur and a formal charge of −1 on the nitrogen. The parent compound is sulfilimine H2S=NH, which is mainly of theoretical interest.

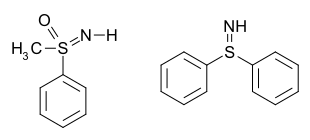
Examples include S,S-diphenylsulfilimine[2] and sulfoximines [Category] such as methylphenylsulfoximine:[3] In the case of a sulfoximine, the bonds can be considered single bonds, with formal charges of −1 on both the oxygen and the nitrogen, and a formal charge of +2 on the sulfur.
Most sulfilimines are N-substituted with electron-withdrawing groups. These compounds are typically prepared by oxidation of thioethers with electrophilic amine reagents, such as chloramine-T in the presence of a base:[4]
An alternative route involves reactions of electrophilic sulfur compounds with amines. The imidosulfonium reagents provide a source of "Me2S2+", which are attacked by amines.
Sulfilimine bonds stabilize collagen IV strands found in the extracellular matrix[5] and arose at least 500 mya.[6] These bonds covalently connect hydroxylysine and methionine residues of adjacent polypeptide strands to form a larger collagen trimer.
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.