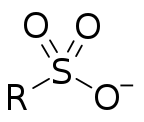
Sulfonate
Organosulfur compound of the form R–S(=O)2–O (charge –1) / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Sulfonate?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
In organosulfur chemistry, a sulfonate is a salt, anion or ester of a sulfonic acid. Its formula is R−S(=O)2−O−, containing the functional group −S(=O)2−O−, where R is typically an organyl group, amino group or a halogen atom. Sulfonates are the conjugate bases of sulfonic acids. Sulfonates are generally stable in water, non-oxidizing, and colorless. Many useful compounds and even some biochemicals feature sulfonates.