
Trazodone
Antidepressant medication / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Trazodone?
Summarize this article for a 10 year old
Trazodone, sold under many brand names,[1] is an antidepressant medication.[20] It is used to treat major depressive disorder, anxiety disorders, and insomnia.[20] The medication is taken orally.[20]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Desyrel, Trittico, others[1] |
| Other names | AF-1161 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a681038 |
| License data | |
| Dependence liability | None[2] |
| Addiction liability | None[2] |
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | By mouth: 65%[7][failed verification] |
| Protein binding | 89–95%[8] |
| Metabolism | Liver (CYP3A4, CYP2D6, CYP1A2?)[9][10][11][12][13] |
| Metabolites | mCPPTooltip meta-Chlorophenylpiperazine[14] |
| Onset of action | By mouth: 1 hour (Tmax)[15] |
| Elimination half-life | • Trazodone (IR): 4–15 hours[9][11][16][17] • Trazodone (ER): 9–13 hours[18][11][16] • mCPPTooltip meta-Chlorophenylpiperazine: 3–16 hours[9][11][17][19] |
| Excretion | Urine: 70–75%[7] Feces: 21%[7] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.039.364 |
| Chemical and physical data | |
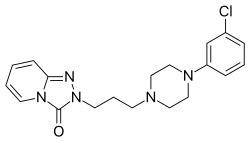
| Formula | C19H22ClN5O |
| Molar mass | 371.87 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 87 °C (189 °F) |
| |
| |
| (verify) | |
Common side effects include dry mouth, feeling faint, vomiting, and headache.[20] More serious side effects may include suicide, mania, irregular heart rate, and pathologically prolonged erections.[20] It is unclear if use during pregnancy or breastfeeding is safe.[21] It is a phenylpiperazine compound of the serotonin antagonist and reuptake inhibitor (SARI) class.[22][23] Trazodone also has sedating effects.[24]
Trazodone was approved for medical use in the United States in 1981.[20] It is available as a generic medication.[20] In 2021, it was the 22nd most commonly prescribed medication in the United States, with more than 26 million prescriptions.[25][26]