
Trimethylgallium
Chemical compound / From Wikipedia, the free encyclopedia
Trimethylgallium, often abbreviated to TMG or TMGa, is the organogallium compound with the formula Ga(CH3)3. It is a colorless, pyrophoric liquid.[1] Unlike trimethylaluminium, TMG adopts a monomeric structure.[2] When examined in detail, the monomeric units are clearly linked by multiple weak Ga---C interactions, reminiscent of the situation for trimethylindium.[3]
Quick Facts Names, Identifiers ...
 | |
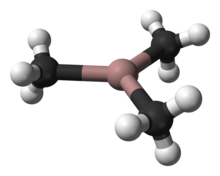 | |
| Names | |
|---|---|
| IUPAC name
trimethylgallane, trimethanidogallium | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.014.452 |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| Ga(CH3)3 | |
| Molar mass | 114.827 g/mol |
| Appearance | colourless liquid |
| Melting point | −15 °C (5 °F; 258 K) |
| Boiling point | 55.7 °C (132.3 °F; 328.8 K) |
| Reacts with water | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
Pyrophoric (can ignite spontaneously in air), reacts with water to release methane |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close