
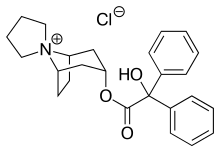
Trospium chloride
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Trospium chloride?
Summarize this article for a 10 year old
Trospium chloride is a muscarinic antagonist used to treat overactive bladder.[2] It has side effects typical of this class of drugs, namely dry mouth, stomach upset, and constipation; these side effects cause problems with people taking their medicine as directed. However it doesn't cause central nervous system side effects like some other muscarinic antagonists.[3] It is in pregnancy category C and is excreted in breast milk.[4]
 | |
| Clinical data | |
|---|---|
| Trade names | Regurin, Sanctura, many generics[1] |
| AHFS/Drugs.com | Monograph |
| Pregnancy category |
|
| Routes of administration | By mouth (tablets, capsules) |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | 50–85% |
| Elimination half-life | 20 hours |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider |
|
| UNII | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.030.784 |
| Chemical and physical data | |
| Formula | C25H30ClNO3 |
| Molar mass | 427.97 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Chemically it is a quaternary ammonium cation which causes it to stay in periphery rather than crossing the blood–brain barrier.[5] It works by causing the smooth muscle in the bladder to relax.[2]
It was patented in 1966 and approved for medical use in 1974.[6] It was first approved in the US in 2004, and an extended release version with once-daily dosing was brought to market in 2007. It became generic in Europe in 2009 and in the US the first extended-release generic was approved in 2012.