User:Aherthabey/sandbox
Chemical compound / From Wikipedia, the free encyclopedia
Sodium iron oxide is an inorganic compound with the formula NaFeO
2. It is a gray, red-grey, brown or black solid that is insoluble in water. The material has attracted attention as a component of sodium-ion batteries[1]. This battery chemistry is targeted for use in power tools, electric vehicles, solar energy installations[2] and more recently large grid-scale energy storage.[3]
 | |
 | |
| Names | |
|---|---|
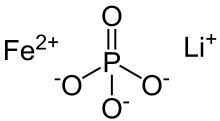
| IUPAC name
iron(2+) lithium phosphate (1:1:1) | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider |
|
| EC Number |
|
PubChem CID |
|
| |
| |
| Properties | |
| FeLiO 4P | |
| Molar mass | 157.757 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Most lithium batteries (Li-ion) used in consumer electronics products use cathodes made of lithium compounds such as lithium cobalt oxide (LiCoO
2), lithium manganese oxide (LiMn
2O
4), and lithium nickel oxide (LiNiO
2). The anodes are generally made of graphite.
Lithium iron phosphate exists naturally in the form of the mineral triphylite, but this material has insufficient purity for use in batteries.