User:Mr. Ibrahem/Fondaparinux
From Wikipedia, the free encyclopedia
Fondaparinux, sold under the brand name Arixtra, is an anticoagulant used to treat and prevent blood clots (deep vein, pulmonary, and superficial vein thrombosis) and to treat unstable angina and heart attacks.[1][3] It is given by injection under the skin.[3]
 | |
| Clinical data | |
|---|---|
| Trade names | Arixtra |
| Other names | Fondaparinux sodium |
| AHFS/Drugs.com | Monograph |
| License data |
|
| Routes of administration | Subcutaneous |
| Drug class | Factor Xa inhibitor[1] |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | N/A |
| Protein binding | 94% |
| Metabolism | renally excreted unchanged |
| Elimination half-life | 17-21 hours[2] |
| Chemical and physical data | |
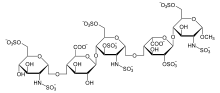
| Formula | C31H43N3Na10O49S8 |
| Molar mass | 1728.03 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Common side effects include bleeding.[1] Other side effects may include low platelets.[1] Use is not recommended in those with serious kidney problems.[3] There is no evidence of harm to the baby with use in pregnancy.[1] It works by blocking factor Xa.[3]
Fondaparinux was approved for medical use in the United States in 2001 and Europe in 2002.[1][3] It is available as a generic medication.[4] In the United Kingdom 2.5 mg costs the NHS about £6.[4] In the United States this amount costs about 20 USD.[5]