
Rolapitant
Pharmaceutical drug / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Varubi?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Rolapitant (INN,[2] trade name Varubi /vəˈruːbi/ və-ROO-bee in the US and Varuby in the European Union) is a drug originally developed by Schering-Plough and licensed for clinical development by Tesaro, which acts as a selective NK1 receptor antagonist (antagonist for the NK1 receptor).[3] It has been approved as a medication for the treatment of chemotherapy-induced nausea and vomiting (CINV) after clinical trials showed it to have similar or improved efficacy and some improvement in safety over existing drugs for this application.[4][5][6][7]
Quick Facts Clinical data, Pronunciation ...
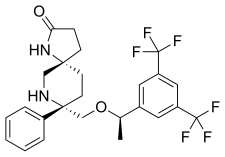 | |
| Clinical data | |
|---|---|
| Pronunciation | /roʊˈlæpɪtænt/ roh-LAP-i-tant |
| Trade names | Varubi (US), Varuby (EU) |
| Other names | SCH 619734 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a615041 |
| License data |
|
| Routes of administration | By mouth (tablets), intravenous |
| Drug class | NK1 receptor antagonists, antiemetics |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | nearly 100% |
| Protein binding | 99.8% |
| Metabolism | CYP3A4 |
| Metabolites | C4-pyrrolidine-hydroxylated rolapitant (major) |
| Elimination half-life | 169–183 hours |
| Excretion | Feces (52–89%), urine (9–20%)[1] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank |
|
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.243.022 |
| Chemical and physical data | |
| Formula | C25H26F6N2O2 |
| Molar mass | 500.485 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Close