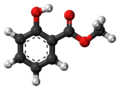
Methyl salicylate
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Wintergreen oil?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Methyl salicylate (oil of wintergreen or wintergreen oil) is an organic compound with the formula C8H8O3. It is the methyl ester of salicylic acid. It is a colorless, viscous liquid with a sweet, fruity odor reminiscent of root beer (in which it is used as a flavoring),[4] but often associatively called "minty", as it is an ingredient in mint candies.[5] It is produced by many species of plants, particularly wintergreens. It is also produced synthetically, used as a fragrance and as a flavoring agent.
Quick Facts Names, Identifiers ...
 | |||
| |||
 | |||
| Names | |||
|---|---|---|---|
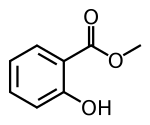
| Preferred IUPAC name
Methyl 2-hydroxybenzoate | |||
Other names
| |||
| Identifiers | |||
| |||
3D model (JSmol) |
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider |
| ||
| ECHA InfoCard | 100.003.925 | ||
| EC Number |
| ||
| KEGG |
| ||
PubChem CID |
|||
| RTECS number |
| ||
| UNII | |||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| C8H8O3 | |||
| Molar mass | 152.149 g/mol | ||
| Appearance | Colorless liquid | ||
| Odor | Sweet, rooty | ||
| Density | 1.174 g/cm3 | ||
| Melting point | −8.6 °C (16.5 °F; 264.5 K) | ||
| Boiling point | 222 °C (432 °F; 495 K)[1] Decomposes at 340–350 °C[2] | ||
| 0.639 g/L (21 °C) 0.697 g/L (30 °C)[2] | |||
| Solubility | Miscible in organic solvents | ||
| Solubility in acetone | 10.1 g/g (30 °C)[2] | ||
| Vapor pressure | 1 mmHg (54 °C)[1] | ||
| Acidity (pKa) | 9.8[3] | ||
| −8.630×10−5 cm3/mol | |||
Refractive index (nD) |
1.538 | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards |
Harmful | ||
| GHS labelling: | |||
 [1] [1] | |||
| Warning | |||
| H302[1] | |||
| P264, P270, P280, P301+P312, P302+P352, P305+P351+P338, P321, P330, P332+P313, P337+P313, P362, P501 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | 96 °C (205 °F; 369 K)[1] | ||
| 452.7 °C (846.9 °F; 725.8 K)[1] | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Close