Top Qs
Timeline
Chat
Perspective
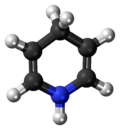
1,4-Dihydropyridine
Chemical compound From Wikipedia, the free encyclopedia
Remove ads
1,4-Dihydropyridine (DHP) is an organic compound with the formula CH2(CH=CH)2NH. The parent compound is uncommon,[2] but derivatives of 1,4-dihydropyridine are important commercially and biologically. The pervasive cofactors NADH and NADPH are derivatives of 1,4-dihydropyridine. Dihydropyridine calcium channel blockers are a class of L-type calcium channel blockers used in the treatment of hypertension. 1,2-Dihydropyridines are also known.[3][4]
Remove ads
Properties and reactions
A recurring feature of 1,4-dihydropyridines is the presence of substituents at the 2- and 6-positions. Dihydropyridines are enamines, which otherwise tend to tautomerize or hydrolyze.[citation needed]
The dominant reaction of dihydropyridines is their ease of oxidation. In the case of dihydropyridines with hydrogen as the substituent on nitrogen, oxidation yields pyridines:
- CH2(CH=CR)2NH → C5H3R2N + H2
The naturally-occurring dihydropyridines NADH and NADPH contain N-alkyl groups. Therefore, their oxidation does not yield pyridine, but N-alkylpyridinium cations:
- CH2(CH=CR)2NR' → C5H3R2NR' + H−
Remove ads
Hantzsch ester

See also
References
External links
Wikiwand - on
Seamless Wikipedia browsing. On steroids.
Remove ads