
Betulinic acid
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about betulinic acid?
Summarize this article for a 10 year old
Betulinic acid is a naturally occurring pentacyclic triterpenoid which has antiretroviral, antimalarial, and anti-inflammatory properties, as well as a more recently discovered potential as an anticancer agent, by inhibition of topoisomerase.[1] It is found in the bark of several species of plants, principally the white birch (Betula pubescens)[2] from which it gets its name, but also the ber tree (Ziziphus mauritiana), selfheal (Prunella vulgaris), the tropical carnivorous plants Triphyophyllum peltatum and Ancistrocladus heyneanus, Diospyros leucomelas, a member of the persimmon family, Tetracera boiviniana, the jambul (Syzygium formosanum),[3] flowering quince (Pseudocydonia sinensis, former Chaenomeles sinensis KOEHNE),[4] rosemary,[5] and Pulsatilla chinensis.[6]
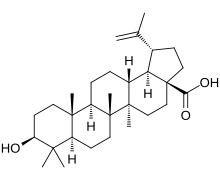 | |
| Names | |
|---|---|
| IUPAC name
3β-Hydroxylup-20(29)-en-28-oic acid | |
| Systematic IUPAC name
(1R,3aS,5aR,5bR,7aR,9S,11aR,11bR,13aR,13bR)-9-Hydroxy-5a,5b,8,8,11a-pentamethyl-1-(prop-1-en-1-yl)icosahydro-3aH-cyclopenta[a]chrysene-3a-carboxylic acid | |
| Other names
Betulic acid Mairin | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider |
|
| ECHA InfoCard | 100.006.773 |
| EC Number |
|
PubChem CID |
|
| UNII | |
| |
| |
| Properties | |
| C30H48O3 | |
| Molar mass | 456.711 g·mol−1 |
| Melting point | 316 to 318 °C (601 to 604 °F; 589 to 591 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |