Pitolisant
Medication to treat narcolepsy / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about pitolisant?
Summarize this article for a 10 year old
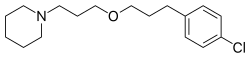
Pitolisant, sold under the brand name Wakix among others, is a medication used for the treatment of excessive daytime sleepiness in adults with narcolepsy.[3] It is a histamine 3 (H3) receptor antagonist/inverse agonist (an antihistamine drug specific to that kind of receptors).[3] It represents the first commercially available medication in its class, so that the US Food and Drug Administration (FDA) declares it a first-in-class medication.[7][8] Pitolisant enhances the activity of histaminergic neurons in the brain that function to improve a person's wakefulness.[9] It was approved by the European Medicines Agency (EMA) in March 2016 for narcolepsy with or without cataplexy, and for excessive daytime sleepiness by the FDA in August 2019.[10] The most common side effects include difficulty sleeping, nausea, and feeling worried.[11]
 | |
| Clinical data | |
|---|---|
| Pronunciation | /pɪˈtɒlɪsənt/ pi-TOL-i-sənt |
| Trade names | Wakix, Ozawade |
| Other names | Tiprolisant; Ciproxidine; BF2.649 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a619055 |
| License data |
|
| Routes of administration | By mouth |
| Drug class | Histamine H3 receptor inverse agonists |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| DrugBank |
|
| ChemSpider | |
| UNII |
|
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C17H26ClNO |
| Molar mass | 295.85 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |