Loading AI tools
Chemical compound From Wikipedia, the free encyclopedia
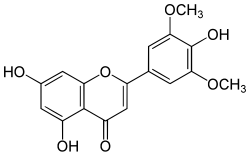
Tricin is a chemical compound. It is an O-methylated flavone, a type of flavonoid. It can be found in rice bran[1] and sugarcane.[2]
 | |
 | |
| Names | |
|---|---|
| IUPAC name
4′,5,7-Trihydroxy-3′,5′-dimethoxyflavone | |
| Systematic IUPAC name
5,7-Dihydroxy-2-(4-hydroxy-3,5-dimethoxyphenyl)-4H-1-benzopyran-4-one | |
| Other names
Tricetin 3′,5′-dimethyl ether | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C17H14O7 | |
| Molar mass | 330.29 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
The biosynthesis of flavones has not yet been elucidated in full; however, most of the mechanistic and enzymatic steps have been discovered and studied. In biosynthesizing tricin, there is first stepwise addition of malonyl-CoA via the polyketide pathway and p-coumaroyl-CoA via the phenylpropanoid pathway.[3] These additions are mediated by the sequential action of chalcone synthase and chalcone isomerase to yield naringenin chalcone and the flavanone, naringenin, respectively. CYP93G1 of the CYP450 superfamily in rice then desaturates naringenin into apigenin. After this step, it is proposed that flavonoid 3',5'-hydroxylase (F3',5'H) changes apigenin into tricetin.[4] Upon formation of tricetin, 3'-O-methyltransferase and 5'-O-methyltransferase add methoxy groups to tricetin to form tricin.

Three flavonolignans derived from tricin have been isolated from oats Avena sativa.[5]
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.