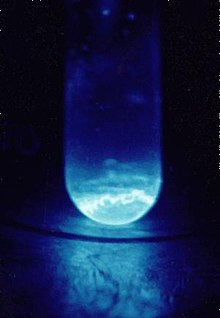
 |
|
| Pronunciation | (eyen-STY-nee-əm) |
|---|
| Appearance | silver-colored |
|---|
| Mass number | [252] |
|---|
|
|
|
| Group | మూస:Infobox element/symbol-to-group/format |
|---|
| Period | period 7 |
|---|
| Block | f-block |
|---|
| Electron configuration | [Rn] 5f11 7s2 |
|---|
| Electrons per shell | 2, 8, 18, 32, 29, 8, 2 |
|---|
|
| Phase at STP | solid |
|---|
| Melting point | 1133 K (860 °C, 1580 °F) |
|---|
| Boiling point | 1269 K (996 °C, 1825 °F) (estimated) |
|---|
| Density (near r.t.) | 8.84 g/cm3 |
|---|
|
| Oxidation states | +2, +3, +4 |
|---|
| Electronegativity | Pauling scale: 1.3 |
|---|
| Ionization energies | |
|---|
 Spectral lines of einsteinium Spectral lines of einsteinium |
|
| Natural occurrence | synthetic |
|---|
| Crystal structure | face-centered cubic (fcc) |
|---|
| Magnetic ordering | paramagnetic |
|---|
| CAS Number | 7429-92-7 |
|---|
|
| Naming | after Albert Einstein |
|---|
| Discovery | Lawrence Berkeley National Laboratory (1952) |
|---|
|
| Template:infobox einsteinium isotopes does not exist |
 Category: Einsteinium Category: Einsteinium
| references |