User:DoroWolf/沙盒/硫酸亚铁
維基百科,自由的 encyclopedia
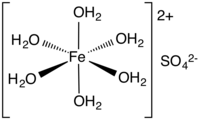
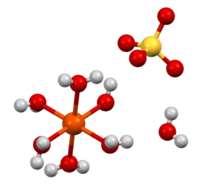
Iron(II) sulfate (British English: iron(II) sulphate) or ferrous sulfate denotes a range of salts with the formula Fe SO4·xH2O. These compounds exist most commonly as the heptahydrate (x = 7) but several values for x are known. The hydrated form is used medically to treat iron deficiency, and also for industrial applications. Known since ancient times as copperas and as green vitriol (vitriol is an archaic name for sulfate), the blue-green heptahydrate (hydrate with 7 molecules of water) is the most common form of this material. All the iron(II) sulfates dissolve in water to give the same aquo complex [Fe(H2O)6]2+, which has octahedral molecular geometry and is paramagnetic. The name copperas dates from times when the copper(II) sulfate was known as blue copperas, and perhaps in analogy, iron(II) and zinc sulfate were known respectively as green and white copperas.[13]
| Iron(II) sulfate | |
|---|---|
 | |
 | |
 | |
| IUPAC名 Iron(II) sulfate | |
| 别名 | Iron(II) sulphate; Ferrous sulfate, Green vitriol, Iron vitriol, Ferrous vitriol, Copperas, Melanterite, Szomolnokite, |
| 识别 | |
| CAS号 | 7720-78-7 17375-41-6 10028-21-4 7782-63-0 |
| PubChem | 24393 62712 62662 |
| ChemSpider | 22804, 56459, 22804 |
| SMILES |
|
| InChI |
|
| InChIKey | BAUYGSIQEAFULO-NUQVWONBAS |
| UN编号 | 3077 |
| ChEBI | 75832 |
| RTECS | NO8500000 (anhydrous) NO8510000 (heptahydrate) |
| 性质 | |
| 化学式 | FeSO4 |
| 摩尔质量 | 151.91 g/mol (anhydrous) 169.93 g/mol (monohydrate) 241.99 g/mol (pentahydrate) 260.00 g/mol (hexahydrate) 278.02 g/mol (heptahydrate) g·mol⁻¹ |
| 外观 | White crystals (anhydrous) White-yellow crystals (monohydrate) Blue-green crystals (heptahydrate) |
| 氣味 | Odorless |
| 密度 | 3.65 g/cm3 (anhydrous) 3 g/cm3 (monohydrate) 2.15 g/cm3 (pentahydrate)[1] 1.934 g/cm3 (hexahydrate)[2] 1.895 g/cm3 (heptahydrate)[3] |
| 熔点 | 680 °C(953 K) |
| 溶解性(水) | Monohydrate: 44.69 g/100 mL (77 °C) 35.97 g/100 mL (90.1 °C) Heptahydrate: 15.65 g/100 mL (0 °C) 19.986 g/100 mL (10 °C) 29.51 g/100 mL (25 °C) 39.89 g/100 mL (40.1 °C) 51.35 g/100 mL (54 °C)[4] |
| 溶解性 | Negligible in alcohol |
| 溶解性(ethylene glycol) | 6.38 g/100 g (20 °C)[5] |
| 蒸氣壓 | 1.95 kPa (heptahydrate)[6] |
| 磁化率 | 6998124000000000000♠1.24×10−2 cm3/mol (anhydrous) 6998105000000000000♠1.05×10−2 cm3/mol (monohydrate) 6998112000000000000♠1.12×10−2 cm3/mol (heptahydrate)[3] 6998101999999999999♠+10200×10−6 cm3/mol |
| 折光度n D |
1.591 (monohydrate)[7] 1.526–1.528 (21 °C, tetrahydrate)[8] 1.513–1.515 (pentahydrate)[1] 1.468 (hexahydrate)[2] 1.471 (heptahydrate)[9] |
| 结构 | |
| 晶体结构 | Orthorhombic, oP24 (anhydrous)[10] Monoclinic, mS36 (monohydrate)[7] Monoclinic, mP72 (tetrahydrate)[8] Triclinic, aP42 (pentahydrate)[1] Monoclinic, mS192 (hexahydrate)[2] Monoclinic, mP108 (heptahydrate)[3][9] |
| 空间群 | Pnma, No. 62 (anhydrous) [10] C2/c, No. 15 (monohydrate, hexahydrate)[2][7] P21/n, No. 14 (tetrahydrate)[8] P1, No. 2 (pentahydrate)[1] P21/c, No. 14 (heptahydrate)[9] |
| 晶格常数 | a = 8.704(2) Å, b = 6.801(3) Å, c = 4.786(8) Å (293 K, anhydrous)[10] |
| 晶格常数 | α = 90°, β = 90°, γ = 90° |
| 配位几何 | Octahedral (Fe2+) |
| 热力学 | |
| ΔfHm⦵298K | −928.4 kJ/mol (anhydrous)[3] −3016 kJ/mol (heptahydrate)[11] |
| S⦵298K | 107.5 J/mol·K (anhydrous)[3] 409.1 J/mol·K (heptahydrate)[11] |
| 热容 | 100.6 J/mol·K (anhydrous)[3] 394.5 J/mol·K (heptahydrate)[11] |
| 药理学 | |
| ATC代码 | B03AA07(B03) |
| 危险性 | |
GHS危险性符号 [6] [6]
| |
| GHS提示词 | Warning |
| H-术语 | H302, H315, H319[6] |
| P-术语 | P305+351+338[6] |
| NFPA 704 | |
| 致死量或浓度: | |
LD50(中位剂量)
|
237 mg/kg (rat, oral)[12] |
| 相关物质 | |
| 其他阳离子 | Cobalt(II) sulfate Copper(II) sulfate Manganese(II) sulfate Nickel(II) sulfate |
| 相关化学品 | Iron(III) sulfate |
| 若非注明,所有数据均出自标准状态(25 ℃,100 kPa)下。 | |
It is on the World Health Organization's List of Essential Medicines.[14] In 2020, it was the 116th most commonly prescribed medication in the United States, with more than 5 million prescriptions.[15][16]
