
Aspirin
Medication / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Aspirin?
Summarize this article for a 10 year old
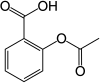
Aspirin, also known as acetylsalicylic acid (ASA), is a nonsteroidal anti-inflammatory drug (NSAID) used to reduce pain, fever, and/or inflammation, and as an antithrombotic.[9] Specific inflammatory conditions which aspirin is used to treat include Kawasaki disease, pericarditis, and rheumatic fever.[9]
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /əˌsiːtəlˌsælɪˈsɪlɪk/ |
| Trade names | Bayer Aspirin, others |
| Other names |
|
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682878 |
| License data | |
| Pregnancy category |
|
| Routes of administration | Oral, rectal |
| Drug class | Nonsteroidal anti-inflammatory drug (NSAID) |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 80–100%[5] |
| Protein binding | 80–90%[6] |
| Metabolism | Liver (CYP2C19 and possibly CYP3A), some is also hydrolysed to salicylate in the gut wall.[6] |
| Elimination half-life | Dose-dependent; 2–3 h for low doses (100 mg or less), 15–30 h for larger doses.[6] |
| Excretion | Urine (80–100%), sweat, saliva, feces[5] |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL |
|
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.000.059 |
| Chemical and physical data | |
| Formula | C9H8O4 |
| Molar mass | 180.159 g·mol−1 |
| 3D model (JSmol) | |
| Density | 1.40 g/cm3 |
| Melting point | 135 °C (275 °F) [8] |
| Boiling point | 140 °C (284 °F) (decomposes) |
| Solubility in water | 3 g/L |
| |
| |
| (verify) | |
Aspirin is also used long-term to help prevent further heart attacks, ischaemic strokes, and blood clots in people at high risk.[9] For pain or fever, effects typically begin within 30 minutes.[9] Aspirin works similarly to other NSAIDs but also suppresses the normal functioning of platelets.[9]
One common adverse effect is an upset stomach.[9] More significant side effects include stomach ulcers, stomach bleeding, and worsening asthma.[9] Bleeding risk is greater among those who are older, drink alcohol, take other NSAIDs, or are on other blood thinners.[9] Aspirin is not recommended in the last part of pregnancy.[9] It is not generally recommended in children with infections because of the risk of Reye syndrome.[9] High doses may result in ringing in the ears.[9]
A precursor to aspirin found in the bark of the willow tree (genus Salix) has been used for its health effects for at least 2,400 years.[10][11] In 1853, chemist Charles Frédéric Gerhardt treated the medicine sodium salicylate with acetyl chloride to produce acetylsalicylic acid for the first time.[12] Over the next 50 years, other chemists, mostly of the German company Bayer, established the chemical structure and devised more efficient production methods.[12]: 69–75
Aspirin is available without medical prescription as a proprietary or generic medication[9] in most jurisdictions. It is one of the most widely used medications globally, with an estimated 40,000 tonnes (44,000 tons) (50 to 120 billion pills)[clarification needed] consumed each year,[10][13] and is on the World Health Organization's List of Essential Medicines.[14] In 2021, it was the 34th most commonly prescribed medication in the United States, with more than 17 million prescriptions.[15][16]