
Chlorophyll a
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Chlorophyll a?
Summarize this article for a 10 year old
Chlorophyll a is a specific form of chlorophyll used in oxygenic photosynthesis. It absorbs most energy from wavelengths of violet-blue and orange-red light, and it is a poor absorber of green and near-green portions of the spectrum.[3] Chlorophyll does not reflect light but chlorophyll-containing tissues appear green because green light is diffusively reflected by structures like cell walls.[4] This photosynthetic pigment is essential for photosynthesis in eukaryotes, cyanobacteria and prochlorophytes because of its role as primary electron donor in the electron transport chain.[5] Chlorophyll a also transfers resonance energy in the antenna complex, ending in the reaction center where specific chlorophylls P680 and P700 are located.[6]
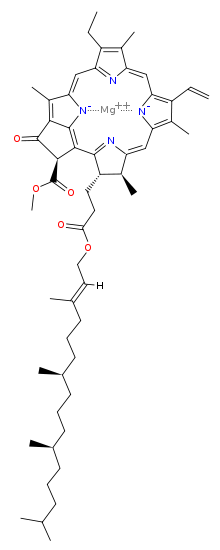 | |
| Names | |
|---|---|
| IUPAC name
Chlorophyll a | |
| Systematic IUPAC name
Magnesium [methyl (3S,4S,21R)-14-ethyl-4,8,13,18-tetramethyl-20-oxo-3-(3-oxo-3-{[(2E,7R,11R)-3,7,11,15-tetramethyl-2-hexadecen-1-yl]oxy}propyl)-9-vinyl-21-phorbinecarboxylatato(2−)-κ2N,N′] | |
| Other names
α-Chlorophyll | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.006.852 |
| EC Number |
|
PubChem CID |
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C55H72MgN4O5 | |
| Molar mass | 893.509 g·mol−1 |
| Appearance | Green |
| Odor | Odorless |
| Density | 1.079 g/cm3[1] |
| Melting point | ~ 152.3 °C (306.1 °F; 425.4 K)[2] decomposes[1] |
| Insoluble | |
| Solubility | Very soluble in ethanol, ether Soluble in ligroin,[2] acetone, benzene, chloroform[1] |
| Absorbance | See text |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |