
Cyclopamine
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Cyclopamine?
Summarize this article for a 10 year old
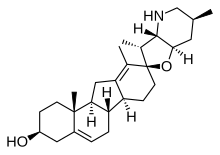
Cyclopamine (11-deoxojervine) is a naturally occurring steroidal alkaloid. It is a teratogenic component of corn lily (Veratrum californicum), which when consumed during gestation has been demonstrated to induce birth defects, including the development of a single eye (cyclopia) in offspring.[1] The molecule was named after this effect, which was originally observed by Idaho lamb farmers in 1957 after their herds gave birth to cycloptic lambs. It then took more than a decade to identify corn lily as the culprit.[2] Later work suggested that differing rain patterns had changed grazing behaviours, which led to a greater quantity of corn lily to be ingested by pregnant sheep.[3] Cyclopamine interrupts the sonic hedgehog signalling pathway, instrumental in early development, ultimately causing birth defects.
 | |
 | |
| Names | |
|---|---|
| IUPAC name
17,23β-Epoxyveratraman-3β-ol | |
| Systematic IUPAC name
(2′R,3S,3′R,3′aS,6′S,6aS,6bS,7′aR,11aS,11bR)-3′,6′,10,11b-Tetramethyl-1,2,3,3′a,4,4′,5′,6,6′,6a,6b,7,7′,7′a,8,11,11a,11b-octadecahydro-3′H-spiro[benzo[a]fluorene-9,2′-furo[3,2-b]pyridin]-3-ol | |
| Other names
• 11-Deoxojervine • (3β,23R)-17,23-Epoxyveratraman-3-ol | |
| Identifiers | |
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider |
|
| ECHA InfoCard | 100.156.363 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C27H41NO2 | |
| Molar mass | 411.630 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |