
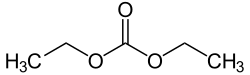
Diethyl carbonate
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Diethyl carbonate?
Summarize this article for a 10 year old
Diethyl carbonate (sometimes abbreviated DEC) is an ester of carbonic acid and ethanol with the formula OC(OCH2CH3)2. At room temperature (25 °C) diethyl carbonate is a colorless liquid with a low flash point.
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Diethyl carbonate | |
| Other names
Carbonic ether; Ethyl carbonate, di-; Eufin[1] | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider |
|
| ECHA InfoCard | 100.003.011 |
| EC Number |
|
PubChem CID |
|
| RTECS number |
|
| UNII | |
| UN number | 2366 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C5H10O3 | |
| Molar mass | 118.132 g·mol−1 |
| Appearance | Colorless liquid |
| Density | 0.975 g/cm3 |
| Melting point | −43 °C (−45 °F; 230 K) [2] |
| Boiling point | 125.9 °C (258.6 °F; 399.0 K) [2] |
| Insoluble | |
| Hazards | |
| GHS labelling: | |
  | |
| Warning | |
| H226, H315, H319, H335 | |
| P210, P233, P240, P241, P242, P243, P261, P264, P271, P280, P302+P352, P303+P361+P353, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P370+P378, P403+P233, P403+P235, P405, P501 | |
| Flash point | 33 °C (91 °F; 306 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Diethyl carbonate is used as a solvent such as in erythromycin intramuscular injections.[3][4][5] It can be used as a component of electrolytes in lithium batteries. It has been proposed as a fuel additive to support cleaner diesel fuel combustion because its high boiling point might reduce blended fuels' volatility, minimizing vapor buildup in warm weather that can block fuel lines.[6] As a fuel additive, it can reduce emissions such as volatile organic compounds, CO2, and particulates.[7]