
Dihydroxymethylidene
Chemical compound / From Wikipedia, the free encyclopedia
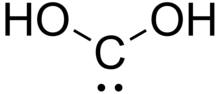
Dihydroxymethylidene or carbonous acid is a chemical compound with formula C(OH)2. It is an unstable tautomer of formic acid. There is no evidence that this compound exists in solution, but the molecule has been detected in the gas phase.[1] Many related carbenes are known, although they are often transient.[2]
Quick Facts Names, Identifiers ...
 | |
 | |
| Names | |
|---|---|
IUPAC name
| |
| Preferred IUPAC name
Dihydroxymethylidene | |
Other names
| |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider |
|
| MeSH | Dihydroxycarbene |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C(OH)2 | |
| Molar mass | 46.025 g·mol−1 |
| Related compounds | |
Related compounds |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close