
Dirhenium decacarbonyl
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Dirhenium decacarbonyl?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
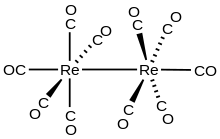
Dirhenium decacarbonyl is the inorganic compound with the chemical formula Re2(CO)10 . Commercially available, it is used as a starting point for the synthesis of many rhenium carbonyl complexes. It was first reported in 1941 by Walter Hieber, who prepared it by reductive carbonylation of rhenium.[2] The compound consists of a pair of square pyramidal Re(CO)5 units joined via a Re-Re bond, which produces a homoleptic carbonyl complex.[3]
Quick Facts Names, Identifiers ...
 | |
 | |
 | |
| Names | |
|---|---|
| IUPAC name
bis(pentacarbonylrhenium)(Re—Re) | |
| Other names
Rhenium carbonyl; rhenium pentacarbonyl | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.034.714 |
| EC Number |
|
PubChem CID |
|
| |
| |
| Properties | |
| Re2(CO)10 | |
| Molar mass | 652.52 g/mol |
| Melting point | 170 °C (338 °F; 443 K) (decomposes) |
| Hazards[1] | |
| GHS labelling: | |
  | |
| Danger | |
| H301, H330, H331, H332 | |
| P261, P271, P304+P340+P311, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close