
Finite subgroups of SU(2)
Use of mathematical groups in magnetochemistry / From Wikipedia, the free encyclopedia
In applied mathematics, finite subgroups of SU(2) are groups composed of rotations and related transformations, employed particularly in the field of physical chemistry. The symmetry group of a physical body generally contains a subgroup (typically finite) of the 3D rotation group. It may occur that the group {±1} with two elements acts also on the body; this is typically the case in magnetism for the exchange of north and south poles, or in quantum mechanics for the change of spin sign. In this case, the symmetry group of a body may be a central extension of the group of spatial symmetries by the group with two elements. Hans Bethe introduced the term "double group" (Doppelgruppe) for such a group, in which two different elements induce the spatial identity, and a rotation of 2π may correspond to an element of the double group that is not the identity.
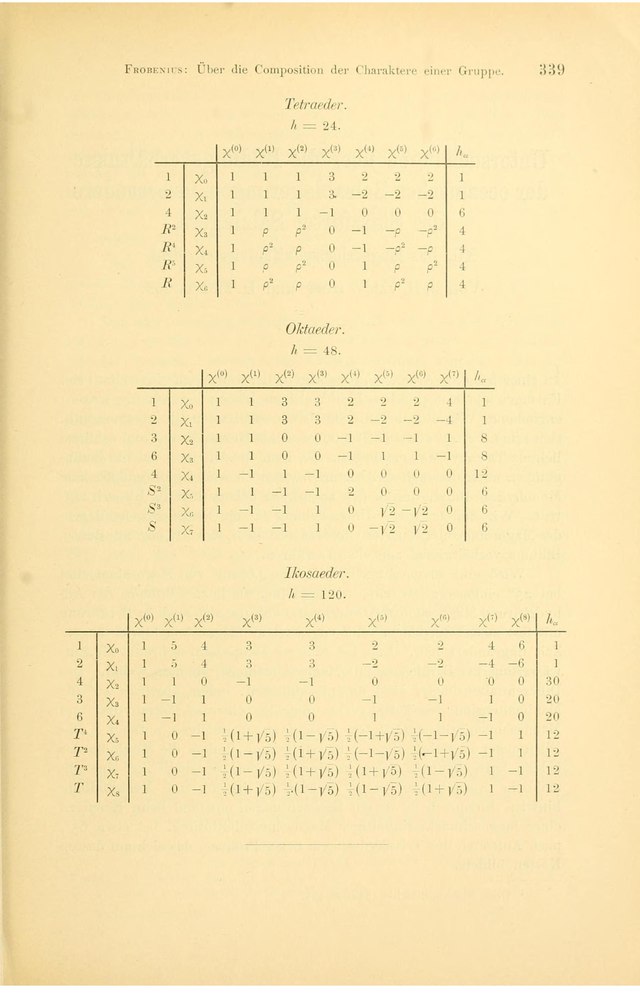
The classification of the finite double groups and their character tables is therefore physically meaningful and is thus the main part of the theory of double groups. Finite double groups include the binary polyhedral groups.
In physical chemistry, double groups are used in the treatment of the magnetochemistry of complexes of metal ions that have a single unpaired electron in the d-shell or f-shell.[2][3] Instances when a double group is commonly used include 6-coordinate complexes of copper(II), titanium(III) and cerium(III). In these double groups rotation by 360° is treated as a symmetry operation separate from the identity operation; the double group is formed by combining these two symmetry operations with a point group such as a dihedral group or the full octahedral group.