
Leucine zipper
DNA-binding structural motif / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Leucine zipper?
Summarize this article for a 10 year old
A leucine zipper (or leucine scissors[1]) is a common three-dimensional structural motif in proteins. They were first described by Landschulz and collaborators in 1988[2] when they found that an enhancer binding protein had a very characteristic 30-amino acid segment and the display of these amino acid sequences on an idealized alpha helix revealed a periodic repetition of leucine residues at every seventh position over a distance covering eight helical turns. The polypeptide segments containing these periodic arrays of leucine residues were proposed to exist in an alpha-helical conformation and the leucine side chains from one alpha helix interdigitate with those from the alpha helix of a second polypeptide, facilitating dimerization.
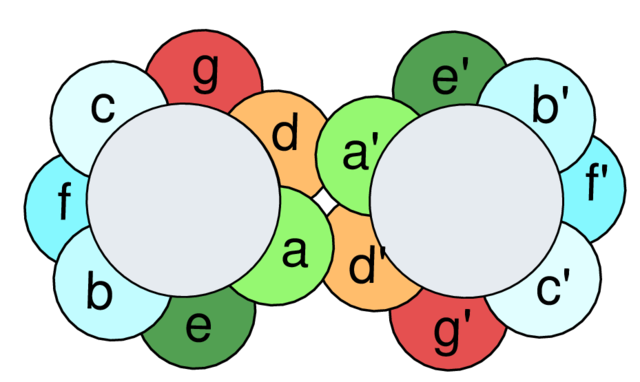
Leucine zippers are a dimerization motif of the bZIP (Basic-region leucine zipper) class of eukaryotic transcription factors.[3] The bZIP domain is 60 to 80 amino acids in length with a highly conserved DNA binding basic region and a more diversified leucine zipper dimerization region.[4] The localization of the leucines are critical for the DNA binding to the proteins. Leucine zippers are present in both eukaryotic and prokaryotic regulatory proteins, but are mainly a feature of eukaryotes. They can also be annotated simply as ZIPs, and ZIP-like motifs have been found in proteins other than transcription factors and are thought to be one of the general protein modules for protein–protein interactions.[5]