
Lubiprostone
Medication used for constipation / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Lubiprostone?
Summarize this article for a 10 year old
Lubiprostone, sold under the brand name Amitiza among others, is a medication used in the management of chronic idiopathic constipation, predominantly irritable bowel syndrome-associated constipation in women and opioid-induced constipation. The drug is owned by Mallinckrodt and is marketed by Takeda Pharmaceutical Company.
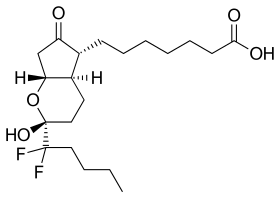 | |
| Clinical data | |
|---|---|
| Trade names | Amitiza |
| Other names | RU-0211 SPI-0211 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a607034 |
| License data |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | Negligible |
| Protein binding | 94% |
| Metabolism | Extensive, CYP not involved |
| Elimination half-life | Unknown (lubiprostone) 0.9–1.4 hours (main metabolite) |
| Excretion | Kidney (60%) and fecal (30%) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.107.168 |
| Chemical and physical data | |
| Formula | C20H32F2O5 |
| Molar mass | 390.468 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
The drug was developed by Sucampo Pharmaceuticals and approved by the Food and Drug Administration (FDA) in 2006.[2][3][4] It was recommended for use in the UK by the National Institute for Health and Care Excellence (NICE) in July 2014.[5] Health Canada approved the drug in 2015.[6] Lubiprostone received approval from the Food and Drug Administration in 2008, to treat irritable bowel syndrome with constipation (IBS-C),[7] and in 2013, for the treatment of opioid-induced constipation in adults with chronic noncancer pain.[4] It is available as a generic medication.[8]