
Nickel(II) chloride
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Nickel(II) chloride?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Nickel(II) chloride (or just nickel chloride) is the chemical compound NiCl2. The anhydrous salt is yellow, but the more familiar hydrate NiCl2·6H2O is green. Nickel(II) chloride, in various forms, is the most important source of nickel for chemical synthesis. The nickel chlorides are deliquescent, absorbing moisture from the air to form a solution. Nickel salts have been shown to be carcinogenic to the lungs and nasal passages in cases of long-term inhalation exposure.[4]
Quick Facts Names, Identifiers ...
 Hexahydrate | |
 Anhydrous | |
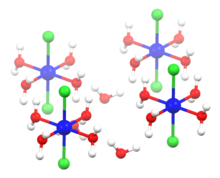 structure of hexahydrate | |
| Names | |
|---|---|
| IUPAC name
Nickel(II) chloride | |
| Other names
Nickelous chloride, nickel(II) salt of hydrochloric acid | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider |
|
| ECHA InfoCard | 100.028.858 |
| EC Number |
|
| KEGG |
|
PubChem CID |
|
| RTECS number |
|
| UNII |
|
| UN number | 3288 3077 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| NiCl2 | |
| Molar mass | 129.5994 g/mol (anhydrous) 237.69 g/mol (hexahydrate) |
| Appearance | yellow-brown crystals deliquescent (anhydrous) green crystals (hexahydrate) |
| Odor | odorless |
| Density | 3.55 g/cm3 (anhydrous) 1.92 g/cm3 (hexahydrate) |
| Melting point | 1,001 °C (1,834 °F; 1,274 K) (anhydrous) 140 °C (hexahydrate) |
| anhydrous 67.5 g/100 mL (25 °C) [1] 87.6 g/100 mL (100 °C) hexahydrate 282.5 g/100 mL (25 °C) [1] 578.5 g/100 mL (100 °C) | |
| Solubility | 0.8 g/100 mL (hydrazine) soluble in ethylene glycol, ethanol, ammonium hydroxide insoluble in ammonia, nitric acid |
| Acidity (pKa) | 4 (hexahydrate) |
| +6145.0·10−6 cm3/mol | |
| Structure | |
| Monoclinic | |
| octahedral at Ni | |
| Thermochemistry | |
Std molar entropy (S⦵298) |
107 J·mol−1·K−1[2] |
Std enthalpy of formation (ΔfH⦵298) |
−316 kJ·mol−1[2] |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
Very toxic (T+) Irritant (Xi) Dangerous for the environment (N) Carcinogen |
| GHS labelling: | |
   | |
| Danger | |
| H301, H315, H317, H331, H334, H341, H350i, H360D, H372, H410 | |
| P201, P202, P260, P261, P264, P270, P271, P272, P273, P280, P281, P285, P301+P310, P302+P352, P304+P340, P304+P341, P308+P313, P311, P314, P321, P330, P332+P313, P333+P313, P342+P311, P362, P363, P391, P403+P233, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
105 mg/kg (rat, oral)[3] |
| Safety data sheet (SDS) | Fischer Scientific |
| Related compounds | |
Other anions |
Nickel(II) fluoride Nickel(II) bromide Nickel(II) iodide |
Other cations |
Palladium(II) chloride Platinum(II) chloride Platinum(II,IV) chloride Platinum(IV) chloride |
Related compounds |
Cobalt(II) chloride Copper(II) chloride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
