
Prucalopride
Drug used to treat chronic constipation / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Prucalopride?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Prucalopride, sold under brand names Resolor and Motegrity among others, is a medication acting as a selective, high affinity 5-HT4 receptor agonist[2] which targets the impaired motility associated with chronic constipation, thus normalizing bowel movements.[3][4][5][6][7][8] Prucalopride was approved for medical use in the European Union in 2009,[9] in Canada in 2011,[10] in Israel in 2014,[11] and in the United States in December 2018.[12] The drug has also been tested for the treatment of chronic intestinal pseudo-obstruction.[13][14]
Quick Facts Clinical data, Trade names ...
 | |
| Clinical data | |
|---|---|
| Trade names | Resolor, Resotran, Motegrity |
| Other names | R-093877, R-108512 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a619011 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank |
|
| ChemSpider | |
| UNII |
|
| KEGG | |
| ChEBI | |
| ChEMBL |
|
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
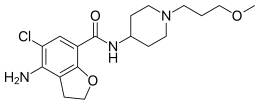
| Formula | C18H26ClN3O3 |
| Molar mass | 367.87 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Close