
Ribokinase
From Wikipedia, the free encyclopedia
In enzymology, a ribokinase (EC 2.7.1.15) is an enzyme that catalyzes the chemical reaction
- ATP + d-ribose ⇌ ADP + d-ribose 5-phosphate
| ribokinase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
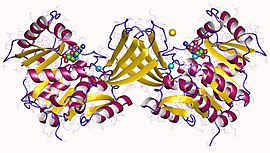 Ribokinase dimer, Human | |||||||||
| Identifiers | |||||||||
| EC no. | 2.7.1.15 | ||||||||
| CAS no. | 9026-84-0 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
Thus, the two substrates of this enzyme are ATP and d-ribose, whereas its two products are ADP and d-ribose 5-phosphate.
The systematic name of this enzyme class is ATP:d-ribose 5-phosphotransferase. Other names in common use include deoxyribokinase, ribokinase (phosphorylating), and d-ribokinase. This enzyme participates in pentose phosphate pathway.
Ribokinase (RK) belongs to the phosphofructokinase B (PfkB) family of sugar kinases.[1] Other members of this family (also known as the RK family) include adenosine kinase (AK), inosine-guanosine kinase, fructokinase, and 1-phosphofructokinase.[1][2][3] The members of the PfkB/RK family are identified by the presence of three conserved sequence motifs and the enzymatic activity of this family of protein generally shows a dependence on the presence of pentavalent ions.[1][2][4] The conserved NXXE motif, which is a distinctive property of the PfkB family of proteins, is involved in pentavalent ion dependency. The structures of RK and several other PfK family of proteins have been determined from a number of organisms.[5] Despite low sequence similarity between AdK and other PfkB family of proteins, these proteins are quite similar at structural levels.[1]