
Sodium aluminate
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Sodium aluminate?
Summarize this article for a 10 year old
Sodium aluminate is an inorganic chemical that is used as an effective source of aluminium hydroxide for many industrial and technical applications. Pure sodium aluminate (anhydrous) is a white crystalline solid having a formula variously given as NaAlO2, NaAl(OH)4 (hydrated),[3] Na2O·Al2O3, or Na2Al2O4. Commercial sodium aluminate is available as a solution or a solid.
Other related compounds, sometimes called sodium aluminate, prepared by reaction of Na2O and Al2O3 are Na5AlO4 which contains discrete AlO45− anions, Na7Al3O8 and Na17Al5O16 which contain complex polymeric anions, and NaAl11O17, once mistakenly believed to be β-alumina, a phase of aluminium oxide.[4][5]
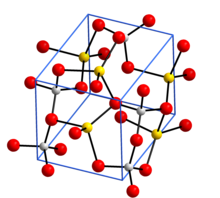 Crystal structure with sodium in yellow, aluminium in grey, and oxygen in red[1] | |
 Sodium metaaluminate sample | |
| Names | |
|---|---|
| IUPAC name
Sodium aluminate | |
| Other names
Sodium aluminium oxide, Sodium metaaluminate Aluminate, ((AlO2)1−), sodium | |
| Identifiers | |
3D model (JSmol) |
|
| ECHA InfoCard | 100.013.728 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| NaAlO2 | |
| Molar mass | 81.97 g/mol |
| Appearance | white powder (sometimes light-yellowish) hygroscopic/ when dissolved in water a colloidal black solution is formed |
| Odor | odorless |
| Density | 1.5 g/cm3 |
| Melting point | 1,650 °C (3,000 °F; 1,920 K) |
| highly soluble | |
| Solubility | Insoluble in alcohol[2] |
Refractive index (nD) |
1.566 |
| Structure | |
| orthorhombic | |
| Thermochemistry | |
Heat capacity (C) |
73.6 J/mol K |
Std molar entropy (S⦵298) |
70.4 J/mol K |
Std enthalpy of formation (ΔfH⦵298) |
-1133.2 kJ/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |