
Titanium butoxide
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Titanium butoxide?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Titanium butoxide is a metal alkoxide with the formula Ti(OBu)4 (Bu = –CH2CH2CH2CH3). It is a colorless odorless liquid although aged samples can appear yellowish. Owing to hydrolysis, samples have a weak alcohol-like odor. It is soluble in many organic solvents.[1][3] Decomposition in water is not hazardous, and therefore titanium butoxide is often used as a liquid source of titanium dioxide, which allows deposition of TiO2 coatings of various shapes and sizes down to the nanoscale.[4][5]
Quick Facts Names, Identifiers ...
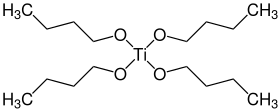 gas phase structure | |
| Names | |
|---|---|
| IUPAC name
Titanium(IV) butoxide | |
| Other names | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.024.552 |
| EC Number |
|
PubChem CID |
|
| UNII | |
| UN number | 2920 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C16H36O4Ti | |
| Molar mass | 340.32164 |
| Appearance | COLORLESS TO LIGHT-YELLOW LIQUID |
| Odor | weak alcohol-like[1] |
| Density | 0.998 g/cm3[1] |
| Melting point | -55 °C[1] |
| Boiling point | 312 °C[1] |
| decomposes[1] | |
| Solubility | most organic solvents except ketones[1] |
Refractive index (nD) |
1.486[1] |
| Thermochemistry | |
Heat capacity (C) |
711 J/(mol·K)[2] |
Std enthalpy of formation (ΔfH⦵298) |
-1670 kJ/mol[2] |
| Hazards | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
3122 mg/kg (rat, oral) and 180 mg/kg (mouse, intravenal).[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
Titanium butoxide is often used to prepare titanium oxide materials and catalysts.[6][7][citation needed]