Titanium ethoxide
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Titanium ethoxide?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Titanium ethoxide is a chemical compound with the formula Ti4(OCH2CH3)16. It is a commercially available colorless liquid that is soluble in organic solvents but hydrolyzes readily. Its structure is more complex than suggested by its empirical formula. Like other alkoxides of titanium(IV) and zirconium(IV), it finds used in organic synthesis and materials science.[1]
Quick Facts Names, Identifiers ...
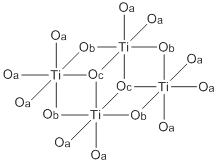 Abbreviated structure of titanium(IV) ethoxide tetramer. The ethoxide ligands are represented by O's. The terminal ethoxide ligands are designated by Oa, the doubly bridging ligands by Ob, and the triply bridging ligands by Oc. | |
| Names | |
|---|---|
| IUPAC name
ethanolate; titanium(4+) | |
| Preferred IUPAC name
Titanium(IV) ethoxide | |
| Systematic IUPAC name
titanium(4+) tetraethanolate | |
| Other names
Ethyl titanate, tetraethyl titanate | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.019.464 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C32H80O16Ti4 | |
| Molar mass | 228.109 g/mol |
| Appearance | colorless liquid |
| Density | 1.088 |
| Melting point | 54 °C (129 °F; 327 K)[original research?] |
| Boiling point | 150–152 °C (302–306 °F; 423–425 K) (@10 mmHg) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close