
Tricalcium phosphate
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Tricalcium phosphate?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
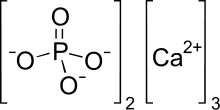
Tricalcium phosphate (sometimes abbreviated TCP), more commonly known as Calcium phosphate, is a calcium salt of phosphoric acid with the chemical formula Ca3(PO4)2. It is also known as tribasic calcium phosphate and bone phosphate of lime (BPL). It is a white solid of low solubility. Most commercial samples of "tricalcium phosphate" are in fact hydroxyapatite.[4][5]
Quick Facts Names, Identifiers ...
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Calcium phosphate | |
| Other names
Tribasic calcium phosphate, tricalcium bis(phosphate) | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider |
|
| ECHA InfoCard | 100.028.946 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| Ca3(PO4)2 | |
| Molar mass | 310.18 g/mol |
| Appearance | White amorphous powder |
| Density | 3.14 g/cm3[1] |
| Melting point | 1,670 °C (3,040 °F; 1,940 K)[1] |
| 1.2 mg/kg[1] | |
Solubility product (Ksp) |
2.07×10−33[2] |
| Thermochemistry | |
Std enthalpy of formation (ΔfH⦵298) |
−4126 kJ/mol (α-form)[3] |
| Pharmacology | |
| A12AA01 (WHO) | |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Related compounds | |
Other anions |
Calcium pyrophosphate |
Other cations |
Trimagnesium phosphate Trisodium phosphate Tripotassium phosphate |
Related compounds |
Monocalcium phosphate Dicalcium phosphate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
It exists as three crystalline polymorphs α, α′, and β. The α and α′ states are stable at high temperatures.
